
This is the reason why H is always a terminal atom and never a central atom. Hydrogen only needs to form one bond to complete a duet of electrons. This octet rule holds for elements in the second and third periods (or rows) of the periodic table. Atom (Group number)īecause hydrogen only needs two electrons to fill its valence shell, it follows the duet rule. This need to gain a filled valance electron shell by having 8 valence electrons is known as the octet rule and explains why certain elements are stable or unstable despite being electrically neutral. Table showing 4 different atoms, each of their number of bonds, and each of their number of lone pairs. In each case, the sum of the number of bonds and the number of lone pairs is 4, which is equivalent to eight (octet) electrons. As we continue through the periodic table in this way, writing the electron configurations of larger and larger atoms, it becomes tedious to keep copying the configurations of the filled inner orbitals. The number of electrons required to obtain an octet determines the number of covalent bonds an atom can form.
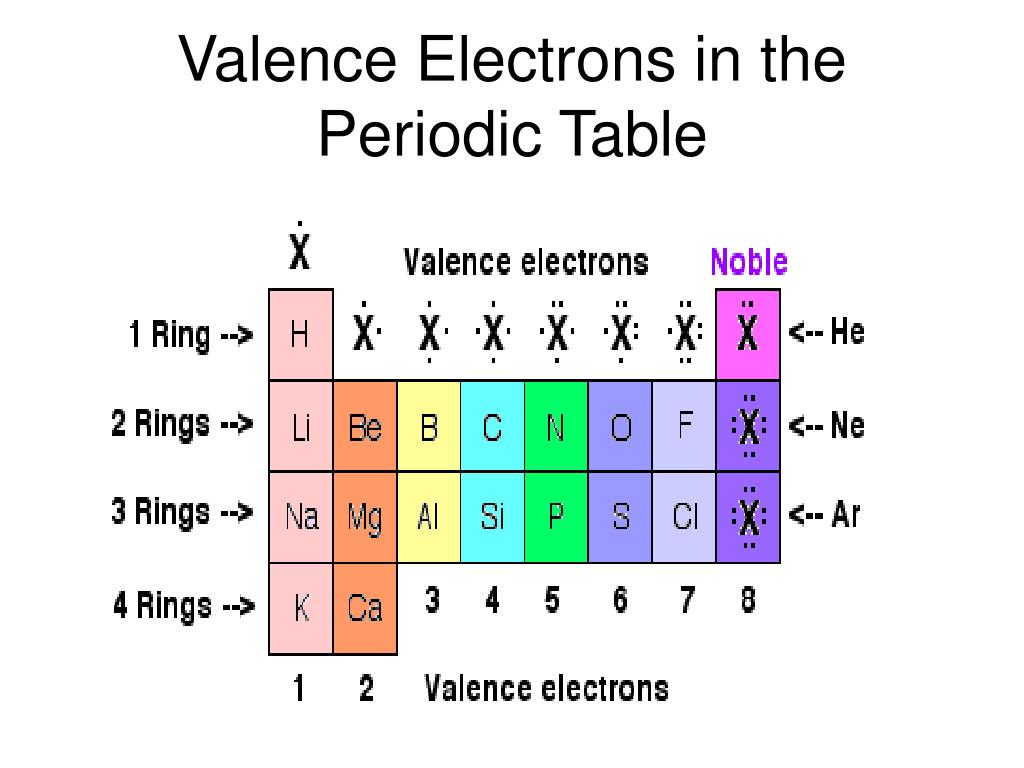
Oxygen and other atoms in group 16 obtain an octet by forming two covalent bonds: To obtain an octet, these atoms form three covalent bonds, as in NH 3 (ammonia). Group 15 elements such as nitrogen have five valence electrons in the atomic Lewis symbol: one lone pair and three unpaired electrons. Label each column on the periodic table of elements from 1 to 18. All of the 1A elements have one valence electron. A group is a vertical column of the periodic table. The number of valence electrons of an element can be determined by the periodic table group (vertical column) in which the element is categorized. The transition elements and inner transition elements also do not follow the octet rule since they have d and f electrons involved in their valence shells. Remember that Mendeleev arranged the periodic table so that elements with the most similar properties were placed in the same group. Because hydrogen only needs two electrons to fill its valence shell, it is an exception to the octet rule and only needs to form one bond. These four electrons can be gained by forming four covalent bonds, as illustrated here for carbon in CCl 4 (carbon tetrachloride) and silicon in SiH 4 (silane). For example, each atom of a group 14 element has four electrons in its outermost shell and therefore requires four more electrons to reach an octet.

The number of valence electrons of an atom can be obtained from the periodic table because it is equal to the group number of the atom. It is said to occupy orbitals in an atom. The paper introduces Chemdex, a freely accessible web-based database of over 70 000 compounds characterised by crystallography from across the periodic table. The number of bonds that an atom can form can often be predicted from the number of electrons needed to reach an octet (eight valence electrons) this is especially true of the nonmetals of the second period of the periodic table (C, N, O, and F). Electrons are involved in the chemical bonding and reactions of the atom. From left to right: water molecule, ammonia molecule, and methane molecule
